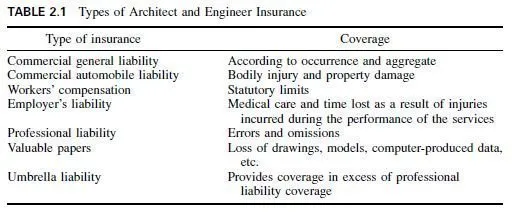
The excellent corrosion resistance of copper makes it suitable for such applications as roofing, flashing, cornices, gutters, downspouts, leaders, fly screens, and similar applications. For roofing and flashing, soft-annealed copper is employed, because it is ductile and can easily be bent into various shapes. For gutters, leaders, downspouts, and similar applications, cold-rolled hard copper is employed, because its greater hardness and stiffness permit it to stand without large numbers of intermediate supports.
Copper and copper-based alloys, particularly the brasses, are employed for water pipe in buildings, because of their corrosion resistance. Electrolytic tough-pitch copper is usually employed for electrical conductors, but for maximum electrical conductivity and weldability, oxygen-free high-conductivity copper is used.
When arsenic is added to copper, it appears to form a tenacious adherent film, which is particularly resistant to pitting corrosion. Phosphorus is a powerful deoxidizer and is particularly useful for copper to be used for refrigerator tubing and other applications where flaring, flanging, and spinning are required. Arsenic and phosphorus both reduce the electrical conductivity of the copper.
For flashing, copper is frequently coated with lead to avoid the green patina formed on copper that is sometimes objectionable when it is washed down over adjacent surfaces, such as ornamental stone. The patina is formed particularly in industrial atmospheres. In rural atmospheres, where industrial gases are absent, the copper normally turns to a deep brown color.
Principal types of copper and typical uses are:
Electrolytic tough pitch (99.90% copper) is used for electrical conductors bus bars, commutators, etc.; building products roofing, gutters, etc.; process equipment kettles, vats, distillery equipment; forgings. General properties are high electrical conductivity, high thermal conductivity, and excellent working ability.
Deoxidized (99.90% copper and 0.025% phosphorus) is used, in tube form, for water and refrigeration service, oil burners, etc.; in sheet and plate form, for welded construction. General properties include higher forming and bending qualities than electrolytic copper. They are preferred for coppersmithing and welding (because of resistance to embrittlement at high temperatures).